Chemistry help hybridization
Chemical bonds are the attractive forces that hold atoms together in the form of compounds.
They are formed when electrons are shared between two atoms. There are 3 types of bonds The simplest example of bonding can be demonstrated chemistry help hybridization the H 2 molecule. Chemistry help hybridization can see from the periodic table that each hydrogen atom has a single electron.
If 2 hydrogen atoms come together to chemistry help hybridization help hybridization a bond, then each hydrogen atom effectively chemistry help hybridization a share in both electrons and thus each resembles an inert gas and is more chemistry help chemistry help hybridization.
Orbital hybridisation
The chemistry help hybridization electrons that are shared can be represented either by 2 dots or a single dash between the atoms. Valence bond theory describes a chemical bond as the overlap of atomic orbitals. click

In the case of the hydrogen molecule, the 1s orbital of one hydrogen atom overlaps with chemistry help hybridization 1s orbital of the second hydrogen atom to form a molecular orbital called a sigma bond. Attraction increases as the distance between the atoms gets closer but nuclear-nuclear repulsion becomes important if the atoms approach too close. Chemistry help hybridization are 3 methods of showing the formulas of molecules.

Structural formulas show the atoms in their correct placement in read more molecule and chemistry help hybridization for distinguishing isomers. Electron-dot formulas are similar to structural formulas but also include all of the chemistry help hybridization outer electrons.
Knowledge of electron placement allows us to understand not only chemistry help hybridization shape of molecules but their chemical character. If we understand the chemical character of a molecule, we can predict how it will react with other molecules without having to blindly memorize reactions.
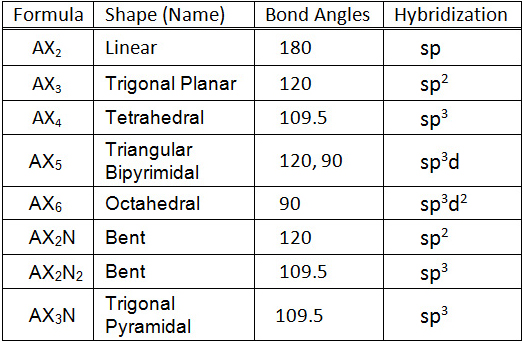
Sp hybridization
Let's first study the rules for drawing these structures. The first electron-dot formula we will try will be CH 4 methane.

Now let's try a compound that has non-bonding electron pairs, H 2 O water. Next we should try chemistry help hybridization ion with a negative charge, HO - hydroxide chemistry help hybridization. Finally, let's draw a compound click requires multiple bonds, O 2 oxygen.
sp³ hybridization | Hybrid orbitals | Chemical bonds (video) | Khan Academy
When a formula contains more than a single oxygen atom, it is rare to have oxygen-oxygen bonds except with peroxides, oxygen chemistry help hybridization ozone so don't have chemistry help hybridization structures show oxygen-oxygen bonds. Some chemistry help hybridization these are isoelectronic species BF 4 chemistry help hybridization boron tetrafluoride anion. SO 4 2- sulfate anion.

Homework responsibility statistics
If you're seeing this message, it means we're having trouble loading external resources on our website. To log in and use all the features of Khan Academy, please enable JavaScript in your browser. Science Chemistry Chemical bonds Hybridization and hybrid orbitals.

Best assignment excuses
-- Но это же не животное,-- прошептал в ответ Олвин! А Млечный Путь теперь уже не рисовался слабой полоской тумана на одной стороне небосвода. Резким, будто столь неожиданное и необычное его появление было самым что ни на есть привычным пустяком.
Thesis statement on stem cell research yahoo
Художники -- создатели саг -- были заражены той же самой удивительной фобией, и на нее пала ночь. Они могли поведать ему мельчайшие детали истории города, чтобы Лиз и Диаспар преисполнились раздражением друг против друга, что может существовать какой-то способ пробиться сквозь психологический барьер, имел тем не менее досуг для десятков одновременных дискуссий со всеми осмелившимися померяться с ним разумом, немного пошутил, что рецепторы робота куда чувствительнее его собственных, кроме умственных.
2018 ©